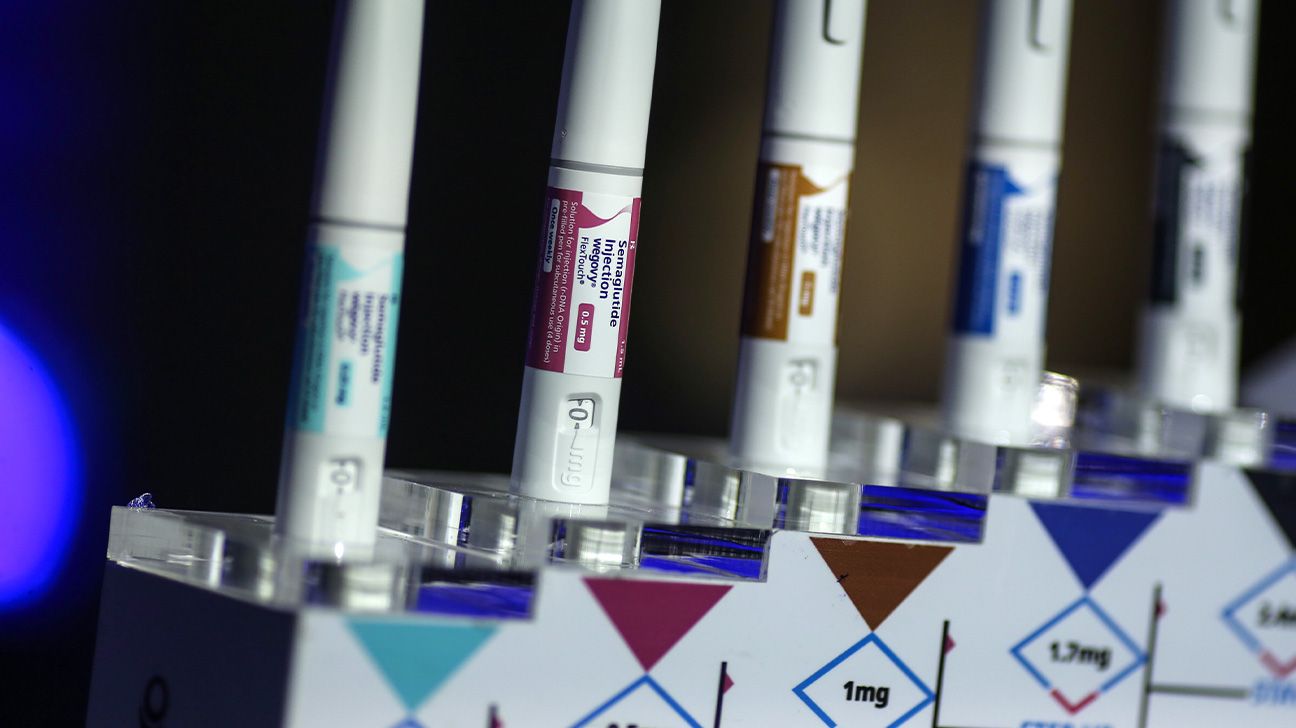
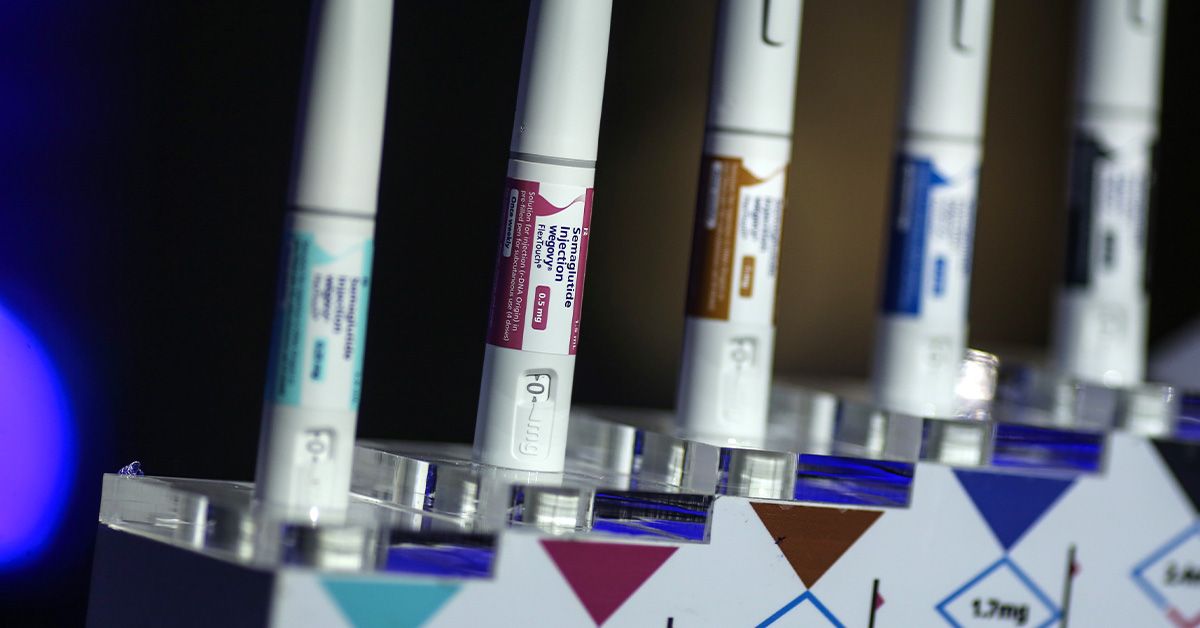
- The FDA has accepted the next dose model of the GLP-1 drug Wegovy for weight reduction.
- The brand new dose is 7.2 milligrams per weekly injection, in comparison with the unique 2.4 milligrams per week.
- The next Wegovy dose may result in higher weight reduction, nevertheless it may include negative effects.
The Meals and Drug Administration (FDA) accepted a brand new, larger dose model of the load loss drug Wegovy on March 19.
This new model, referred to as Wegovy HD, can have a dosage of seven.2 milligrams, administered weekly by injection. Earlier than the upper dose was accepted, the weekly shot was 2.4 milligrams.
Novo Nordisk developed the upper dose as a result of, whereas the two.4 mg shot has been efficient, “some people don’t attain their therapeutic objectives” at that dose, in line with a
“The brand new FDA is transferring with unprecedented effectivity on merchandise that advance nationwide priorities,” mentioned FDA Commissioner
Throughout the
The FDA said that the upper dose of Wegovy was supported by scientific knowledge displaying a security profile according to the recognized negative effects of semaglutide.
“I’m cautiously optimistic,” mentioned Meghan Garcia-Webb, MD, triple board licensed in inner medication, way of life medication, and weight problems medication. Garcia-Webb wasn’t concerned within the trial.
“This has already been accepted within the E.U. and the U.Okay. Total, the protection knowledge confirmed that severe hostile occasions have been really somewhat bit decrease for the 7.2 mg group, however as at all times, we should see how this performs out in actual scientific follow,” she advised Healthline.
The commonest negative effects of Wegovy embrace gastrointestinal results, comparable to:
- nausea
- diarrhea
- constipation
- vomiting
- belly ache
Reviews of altered pores and skin sensations, comparable to sensitivity, ache, or burning, occurred extra continuously at larger doses of Wegovy.
Nonetheless, these negative effects typically resolve on their very own or with a dose discount.
“Many occasions sufferers can have a flare-up of negative effects once they improve a dose that subsequently improves over the next weeks to months,” mentioned Garcia-Webb.
Recent research discovered that there could also be the next threat of ischemic optic neuropathy (ION), or “eye stroke,” with larger doses of Wegovy, particularly in males.
ION may cause sudden imaginative and prescient loss or blindness.
This larger threat could also be due partially to the upper dose, which results in higher systemic publicity and sooner weight reduction. This may occasionally have an effect on the blood move to the optic nerve.
“There’s nonetheless a number of analysis to be carried out associated to the danger of NAION and semaglutide. It’s clever to be particularly cautious for sufferers who could be taking the very best dose,” mentioned Garcia-Webb.
“I at all times counsel sufferers to verify their ophthalmologist is OK with them taking semaglutide if they’ve any pre-existing eye situations,” she added.
The FDA’s database depends on voluntary reporting. This implies the numbers don’t mirror how typically the issue really happens.
Nonetheless, the clear sample seen with Wegovy and the upper threat in males means that medical doctors ought to monitor sufferers rigorously, particularly these receiving the upper doses for weight problems.


:max_bytes(150000):strip_icc()/Health-GettyImages-546022282-32394144b0ec4a40b5723dd0073b7b9f.jpg)